Recently Published Articles

From Discussion to Action: 5 Key Takeaways from the HPRC Spring Meeting
Shahin Sandino
Education
Sustainability
— 2 minute read
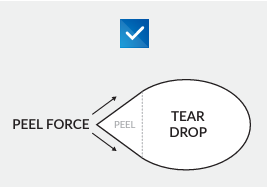
Best Practices for Designing a Die Cut Lid With Your Supplier
Jeremy Elwell
Education
— 2 minute read
Contact Us
CONTACT US
CHAT

.png)
