How Adhesive Coating Can Protect Your Medical Device and the Patient
This article explains how different packaging materials are bonded together in a sterile barrier pouch, the difference between coated and uncoated pouches, and the potential challenges involved in using an uncoated pouch.
Generally, to bond different packaging materials together, an intermediate medium is needed for the transition. This medium is known as the coating or adhesive layer. A coating formula is designed in cohesion-peeling mode, which has a controllable strength so a user can peel it from the middle of the coating layer with his or her hands, with a peelable seal provided.
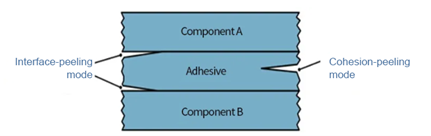
Since a cohesion-peeling mode is used, the coating layer will leave minimal traces on the surface of the two materials, thereby guaranteeing visual integrity of the seal, which may be used as a method for quickly determining whether the sterile barrier is in-tact. In addition, since peeling occurs in the middle of the coating or adhesive layer, the possibility of a fibrous thread of a material being picked up is effectively averted, which achieves a reduction in the risk of fiber tearing.
Let’s look at Tyvek as an example of an uncoated material. First, the material itself is not bondable to another material; second, if it is directly heat-sealed without a coating medium, due to equipment performance and operator proficiency during the sealing process, the package may be over-sealed, resulting in an appearance deformation, edge over-melting or scorching, or serious material delamination upon peeling. Additionally, if the package is weakly sealed, which can cause passages in the heat-sealed edge, the quality of the product can be seriously impacted, and even threaten the life of a patient.

As the most commonly used medical packaging material, commercially available medical sterile pouches comprise both coated and uncoated materials.
Adhesive Coating Can Achieve Higher Product Safety
-
The coating layer, as the intermediate layer, makes the package peelable so that medical staff can quickly and safely open the medical device packaging, especially when opening a long or large package.
-
It is possible to determine the seal integrity of the package through the effect of adhesive transfer, thereby checking that the package integrity has no contamination, so as to ensure the safety of the patient.
-
The coating layer allows the package to quickly achieve an effect of stable sealing, have a stable and uniform peel strength, and an excellent adhesive transfer effect, which prevents abnormal material delamination when the package is opened.
-
A relatively wide variety of sealing process parameters are available, allowing easier operations by equipment and production personnel.
Common Problems Resulting from Uncoated Packaging
-
The seal strength is unstable, which causes inconvenience in operations by production personnel, and instability in the product quality.
-
The problem with the quality of edge sealing increases the difficulty of product verification.
-
The peel strength is unstable, and a layer laceration often occurs when the package is opened, causing secondary particulate contamination.
-
The packaging is not easy to open, which increases the labor intensity of the medical staff.
-
The difficulty of visual inspection increases, which makes it inconvenient to quickly determine whether the seal is in-tact.
In my experience, adding an adhesive coating to your medical packaging is a best practice because it strengthens your packaging, protecting the sterile barrier from a breach. In addition, the adhesive can ensure enhanced peace of mind since validating seal integrity is easier with coated packaging.
Related Regulations:
- YY-T 0681.11-2014 Test Method for Sterile Medical Device Packaging Part 11: Visual Inspection of Sealing Integrity of Medical Packaging;- Article 11.4 of the General Safety and Performance Requirements (GSPR) of the MDR: "It shall be ensured that the integrity of the packaging is clearly evident to the final user"



