Packaging Insights from a Cardiovascular Simulation Center
Surgical simulation centers serve as a critical tool for healthcare professionals, offering a means for education, training, and practice for a variety of surgical scenarios in a realistic operating room environment.
But these centers can also function as an innovative way for others in the healthcare industry to gain important insights into the challenges faced by personnel in the OR, including medical device packaging engineers. I recently interviewed Dr. Robert Cuff, vascular surgeon and former director of the Jacob and Lois Mol Cardiovascular Simulation Center in Grand Rapids, to understand a bit more about the type of information packaging engineers can use to better design with the end user and patient in mind.
HS: Is there anything you have noticed during surgical simulations that you think would be important to consider in terms of packaging design/usability?
RC: The use of a surgical simulation environment provides an excellent opportunity to assess packaging for ease of opening, risk of contamination within the surgical field, and the potential impact of packaging on device storage and organization within the operating room.
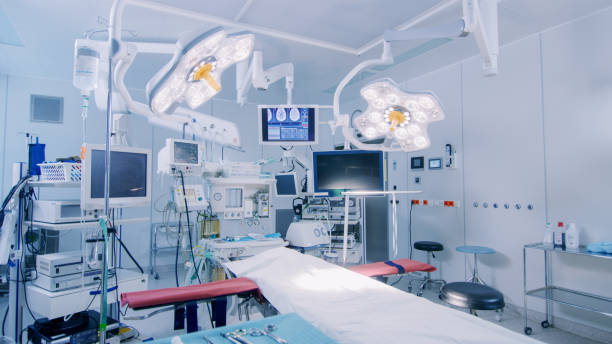
For example, large, bulky packages are often not able to be stored within the operating room theater and would need to be stored in a central storage area. This requires personnel to leave the operating room area to retrieve these items when needed. This can create a delay in the procedure, which has a direct patient impact, as well as become a safety issue when personnel are now out of the operating theatre. A simulated operating room environment can provide this type of insight to packaging professionals. Simulations also allow experienced personnel to assess new types of packaging in a “real life” setting to help them understand and prepare for the impact that PPE has on ease of package opening and allows them to practice minimizing the risk of contamination or loss/drop of a product. Packaging professionals can take advantage of these assessments when designing a package.
HS: Who in the OR is most impacted by packaging design during certain points of a vascular procedure? What matters most to them?
RC: Generally speaking, most packaging is opened during the procedure by the circulating nurse and the scrub tech. The package is usually opened by the surgical nurse, and internal packaging or the product itself is passed to the scrub tech using an aseptic transfer technique. While an internal, sterile package over the product minimizes the chance of contamination by allowing the scrub tech to open the final package, it also increases the amount of packaging needing to be handled and disposed of within the operating room. Seeking a balance between the amount of packaging, ease of opening, and minimizing risk of product being dropped or contaminated is what is needed. Packages that are clearly marked to aid in opening, as well as packaging that can be easily handled while attempting to pass product off to the sterile field, is what is most important to the OR team.
HS: What are some recent shifts or trends you have noticed in devices in the vascular space?
RC: Over the past 20 years, there has been a significant movement away from open surgical procedures to endovascular procedures. Typically, instruments used in open surgical procedures are re-processed and therefore involve large sterilization trays with minimal disposable packaging. In contrast, endovascular procedures are usually performed with disposable, individually wrapped instruments. This has led to a significant increase in the amount of packaging that needs to be opened in the operating room near the sterile field, leading to a greater potential for device contamination as well as a significant increase in the amount of packaging waste created in the OR.
When it comes to healthcare packaging, patient safety is the number one priority, and any opportunity to better understand an actual use case scenario can help positively impact package design as it relates to the patient. If you are a packaging engineer in the healthcare industry, consider partnering with a local simulation center to gain insights that can be fed directly into design inputs when designing a new package or improving an existing one.



